Chi-Med Announces Positive Phase II Proof-of-Concept Data for HMPL-004 in Ulcerative Colitis
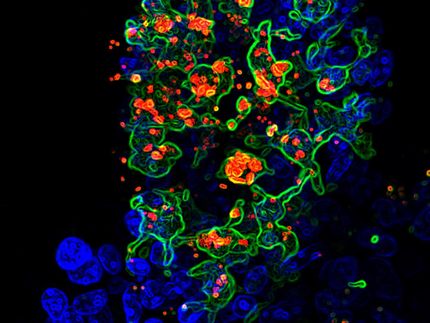
Chi-Med, the Hutchison Whampoa backed pharmaceutical and healthcare Group, announced positive results for its Phase II proof-of-concept study for HMPL-004 in mild-to-moderate Ulcerative Colitis, a form of inflammatory bowel disease. The trial met its objective in that HMPL-004 was well tolerated and showed an equivalent drop in clinical symptom score to the comparator drug, Mesalazine, the current first-line standard of care in mild-to-moderate Ulcerative Colitis.
The Phase II proof-of-concept study, conducted by Chi-Med's wholly-owned drug R&D subsidiary, Hutchison MediPharma Limited, was a multi-center, randomized, double-blind, comparator study of 120 patients with mild-to-moderate Ulcerative Colitis conducted in China. The study evaluated HMPL-004 at 400mg taken three times a day, orally, compared to Mesalazine, the current first-line standard of care. The four trial endpoints were patients: clinical symptom score; overall clinical evaluation; colonoscopic score; and safety evaluation. After treatment for eight weeks, the percentage of patient> '> s clinical symptom score reduction for HMPL-004 was 56% versus 59% for Mesalazine in the Intent-To-Treat population. The overall remission rate (combination of complete and partial remissions) for HMPL-004 was 57% by clinical score compared to 53% for Mesalazine in the Intent-To-Treat population and 47% for HMPL-004 versus 42% for Mesalazine by colonoscopy in the Intent-To-Treat population. HMPL-004 was well tolerated in the study and the adverse event rate was half that of the Mesalazine group
HMPL-004 is an orally active, proprietary botanical product that acts on multiple targets in the pathogenesis of inflammation. It is a compound extracted from a Chinese herb that has extensive history of use in China and South East Asia against respiratory infections and inflammation. This documentation enabled Chi-Med to accelerate the clinical process for HMPL-004 by by-passing Phase I trials, based on the FDA's 2004 guidance on botanical drug products. HMPL-004 is currently also in Phase II trial in the US for the treatment of Crohn's Disease.
Most read news
Topics
Organizations
Other news from the department research and development

Get the life science industry in your inbox
From now on, don't miss a thing: Our newsletter for biotechnology, pharma and life sciences brings you up to date every Tuesday and Thursday. The latest industry news, product highlights and innovations - compact and easy to understand in your inbox. Researched by us so you don't have to.