Cornell's cancer vaccine begins clinical trials
Cornell's cancer vaccine for ovarian cancer and melanoma begins clinical trials
Advertisement
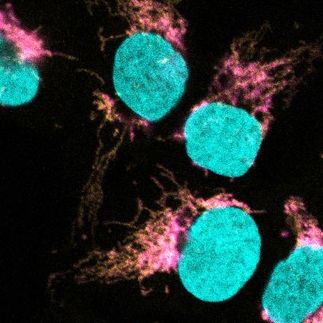
The Bioproduction Facility at Cornell has produced the first batch of a cancer vaccine that is now being used in clinical trials for patients facing either ovarian cancer or melanoma.
The melanoma trial is being conducted at New York University Medical Center, while the ovarian cancer vaccine trial is at the Roswell Park Cancer Institute in Buffalo, N.Y. The trials are assessing the safety and the anti-tumor immune response of the so-called NY-ESO-1 recombinant protein cancer vaccine alone and in combination with other agents, according to the Cancer Research Institute (CRI), an organization that has recently given $450,000 to Cornell to support vaccine production at the Bioproduction Facility. The facility is a partnership between the Ludwig Institute for Cancer Research and Cornell. The goal of these trials is to maximize the body's immune response to the NY-ESO-1 protein.
"Making NY-ESO-1 available for these clinical trials allows investigators to test ways to develop effective cancer vaccines," says Carl A. Batt, Cornell Liberty Hyde Bailey Professor of Food Science and director of the Bioproduction Facility.
"This vaccine is not intended to prevent cancer, but one that will stimulate the body to fight an existing tumor. The challenge is that the vaccine is made from molecules - proteins - that are found in our own bodies and do not normally induce strong immune responses. So part of this trial is to understand how to make the body react to these molecules and attack the cancer."