Luminex Corporation and ViroNovative Announce License Agreement for Human Metapneumovirus Rights
Luminex Corporation and ViroNovative BV announced that the companies have signed a licensing agreement that gives Luminex worldwide rights to use ViroNovative's human metapneumovirus (hMPV) intellectual property in the xTAG Respiratory Viral Panel (RVP). Under the terms of the agreement, rights to ViroNovative's hMPV intellectual property will be extended to Luminex's distributors and end-user customers around the world.
"We are pleased to announce this license agreement in place with ViroNovative for human metapneumovirus," said Jeremy Bridge-Cook, vice president of Luminex Molecular Diagnostics. "This virus is of increasing concern to healthcare providers worldwide. Having this license as part of our xTAG RVP assay is important to us as it allows our customers to use this cutting-edge test without the need to individually secure rights to this intellectual property."
Luminex's xTAG RVP received 510(k) clearance from the U.S. Food and Drug Administration (FDA) on January 3, 2008. The test was cleared for the detection and identification of 12 viruses and viral subtypes that are together responsible for more than 85 percent of respiratory viral infections. xTAG RVP is the first multiplexed nucleic acid test for respiratory viruses cleared for in vitro diagnostic use by the FDA and is the first and only test cleared by FDA for the detection of hMPV.
Other news from the department business & finance

Get the life science industry in your inbox
By submitting this form you agree that LUMITOS AG will send you the newsletter(s) selected above by email. Your data will not be passed on to third parties. Your data will be stored and processed in accordance with our data protection regulations. LUMITOS may contact you by email for the purpose of advertising or market and opinion surveys. You can revoke your consent at any time without giving reasons to LUMITOS AG, Ernst-Augustin-Str. 2, 12489 Berlin, Germany or by e-mail at revoke@lumitos.com with effect for the future. In addition, each email contains a link to unsubscribe from the corresponding newsletter.
Most read news
More news from our other portals
Last viewed contents
Analytica China 2014: Closed with overall success - analytica China further strengthens its position as leading platform
Diosynth Biotechnology enters long-term commercial supply agreement with Dendreon
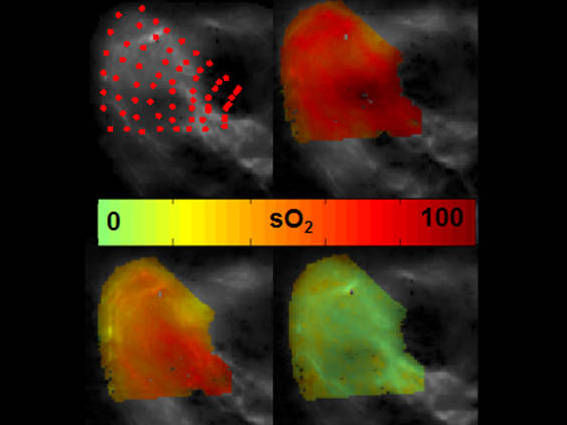
A look beneath the skin - New non-invasive imaging method for showing oxygen in tissue