"Combination" Lyme Disease Vaccine Proteins Patented
Advertisement
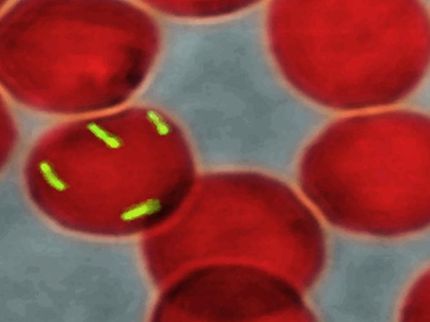
Scientists at the U.S. Department of Energy's Brookhaven National Laboratory and collaborators at Stony Brook University have received U.S. Patent Number 7,179,448 for developing chimeric, or "combination," proteins that may advance the development of vaccines and diagnostic tests for Lyme disease.
The genetically engineered proteins combine pieces of two proteins that are normally present on the surface of the bacterium that causes Lyme disease, but at different parts of the organism's life cycle. "Combining pieces of these two proteins into one chimeric protein should trigger a 'one-two-punch' immune response more capable of fending off the bacterium than either protein alone," says Brookhaven biologist John Dunn, a researcher on the BNL Lyme disease team. "These chimeric proteins could also be used as diagnostic reagents that distinguish disease-causing strains of bacteria from relatively harmless ones, and help assess the severity of an infection," Dunn said.
Scientists have been working on vaccines based on the structures of proteins found on the outer surface of Borrelia burgdoferi, the bacterium that causes Lyme disease. Dunn and colleagues deciphered the atomic level structures of these proteins, known as outer surface proteins A and C (OspA and OspC), at the National Synchrotron Light Source (NSLS) at Brookhaven Lab. The OspA protein, which was used to make the original vaccine against Lyme disease, is only present in the bacteria while they are in the cold-blooded deer tick's stomach, and not in the host. After the tick bites a warm-blooded mammalian host, the injected bacteria produce OspC on their surface.
With the aim of developing a vaccine that would trigger an immune response against both these life cycle stages, Dunn's team used methods of recombinant DNA to create new proteins that combine the most immunogenic portions of OspA and OspC - that is, the regions of the two proteins that are most likely to trigger an immune response.
The researchers have demonstrated that the new combination proteins retain the ability to trigger an immune response to at least one or both of the antigens, and can trigger the production of antibodies that inhibit growth of and/or kill Borrelia bacteria in laboratory cultures. They've also shown that the chimeric proteins can be mass-produced in E. coli bacteria, a common laboratory technique for making proteins, and easily purified for possible use in vaccines or diagnostic assays.
The patent covers the development of the chimeric proteins themselves, the nucleic acids used to generate the proteins, the methods used to make the proteins, the methods used to deliver either the proteins or nucleic acids, the use of the proteins in diagnostic assays or kits, and their use in animals and humans as vaccines against Lyme disease.
Other news from the department research and development
Most read news
More news from our other portals
See the theme worlds for related content
Topic world Antibodies
Antibodies are specialized molecules of our immune system that can specifically recognize and neutralize pathogens or foreign substances. Antibody research in biotech and pharma has recognized this natural defense potential and is working intensively to make it therapeutically useful. From monoclonal antibodies used against cancer or autoimmune diseases to antibody-drug conjugates that specifically transport drugs to disease cells - the possibilities are enormous
Topic world Antibodies
Antibodies are specialized molecules of our immune system that can specifically recognize and neutralize pathogens or foreign substances. Antibody research in biotech and pharma has recognized this natural defense potential and is working intensively to make it therapeutically useful. From monoclonal antibodies used against cancer or autoimmune diseases to antibody-drug conjugates that specifically transport drugs to disease cells - the possibilities are enormous