QIAGEN Significantly Expands its Portfolio of Licenses to real-time PCR
Two New Licenses Added to Technology Portfolio targeting Molecular Diagnostics
QIAGEN N.V. announced that it has acquired licenses from Roche, as well as from Ortho Clinical Diagnostics. According to the company, these two license agreements significantly expand QIAGEN's rights to real-time polymerase chain reaction (PCR) for use in In Vitro Diagnostics (IVD). Financial terms were not disclosed.
The two separate and independent agreements provide QIAGEN with access to a major portfolio of additional patent and marketing rights, which were not covered by the original licensing agreements with Roche. Through these agreements QIAGEN is broadening its technology portfolio to assay technologies and diagnostic tests using almost any basic PCR patent and real-time PCR patent rights related to reagents and methods for practicing PCR and real-time PCR for IVD.
Through the license extension agreement with Roche QIAGEN is now licensed under all real-time polymerase chain reaction (PCR) patents and pending patent applications owned or controlled by Roche Diagnostics, a division of F. Hoffmann-La Roche Ltd, Basel, Switzerland. The agreement with Roche includes, but is not limited to, additional enzyme patent rights further PCR improvement patent rights and pathogen specific patent rights.
In addition, QIAGEN obtained a license from Ortho Clinical Diagnostics to patents related to a taq-polymerase antibody mediated method used to initiate PCR. This is often referred to as a "Hot-Start" technology because it accelerates the activation of PCR enzymes and thereby accelerates the start of the PCR process. The license gives QIAGEN rights in the fields of in research, applied testing and IVD.
Other news from the department research and development
These products might interest you

Micro-Dx™ CE IVD by Molzym
Fully automated from sample to PCR analysis
Rapid identification of bacteria and fungi without time-consuming cultivation
DNA-free Taq Polymerases and Mastermixes by Molzym
DNA-free reagents for unrivalled sensitivity in molecular biology
Purity that makes the difference
DNA/RNA Shield™ SafeCollect Collection Kits by Zymo Research
Sample collection devices for simple & safe at-home testing
Best user experience for secure self-collection
Recombumin® Elite by Sartorius
ICHQ7 cGMP-compliant albumin for biotechnological applications
Increase consistency and safety for gene therapies and vaccines
CellGenix® Growth Factors and Cytokines by Sartorius
Recombinant growth factors without animal products
Optimised cell culture for T cells and MSCs in gene therapy

Greener Alternative Products by Merck Life Science
Sustainable laboratory products for environmentally conscious research
Over 2,500 ecological alternatives to reduce your laboratory footprint

Get the life science industry in your inbox
By submitting this form you agree that LUMITOS AG will send you the newsletter(s) selected above by email. Your data will not be passed on to third parties. Your data will be stored and processed in accordance with our data protection regulations. LUMITOS may contact you by email for the purpose of advertising or market and opinion surveys. You can revoke your consent at any time without giving reasons to LUMITOS AG, Ernst-Augustin-Str. 2, 12489 Berlin, Germany or by e-mail at revoke@lumitos.com with effect for the future. In addition, each email contains a link to unsubscribe from the corresponding newsletter.
More news from our other portals
See the theme worlds for related content
Last viewed contents

Electronic device detects molecules linked to cancer, Alzheimer's and Parkinson's - Inexpensive portable biosensor developed
Cangene's VIG (Vaccinia Immune Globulin Intravenous) contract with U.S. government extended for another five years
Diabetes_mellitus
RETSCH relocates to new, larger premises
Genetic testing for personalized nutrition leads to better outcomes
Seeing more with PET scans - New chemistry for medical imaging
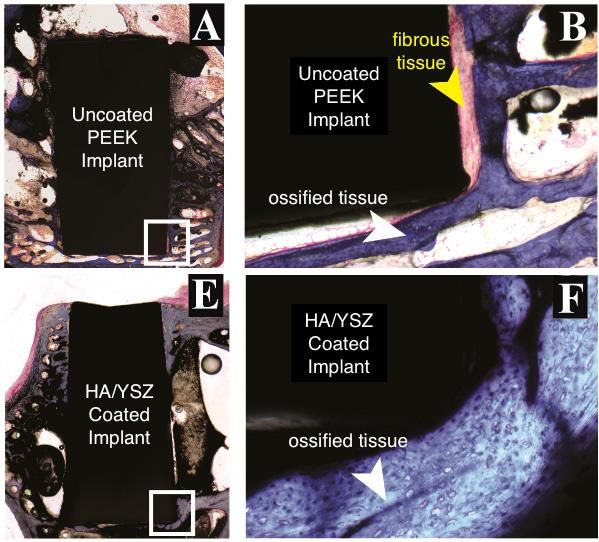
Bioactive film improves how implants bond with bone in animal study
Refractory_period
Promega and Leica Microsystems Sign Agreement to Develop Fluorescent Ligands Enabling Live Cell Imaging at the Sub 100 nm Range

Little Trust in Dr. ChatGPT - There is still a long way to go before people trust medical advice from an AI
UCLA scientists make paralyzed rats walk again after spinal-cord injury