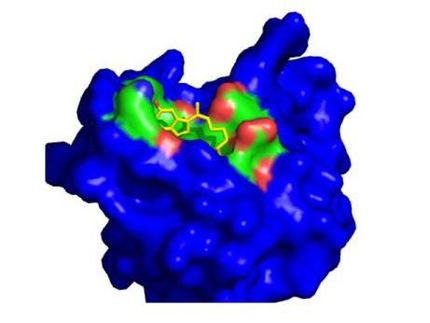
Hybrigenics’ inecalcitol gets Orphan Drug designation for the treatment of Acute Myeloid Leukemia in the United States
Hybrigenics announces the Orphan Drug Designation by the American Food and Drug Administration of inecalcitol for the treatment of acute myeloid leukemia (AML) in the United States. This favourable decision is based on in vitro and in vivo preclinical evidence showing the synergy between inecalcitol and azacytidine or decitabine, two hypo-methylating anticancer drugs, to inhibit the growth of human AML cell lines in vitro and, in vivo, to prolong the survival of mice in two different experimental models of AML.
The molecular basis of their synergy with inecalcitol, a vitamin D receptor agonist, has been elucidated: azacytidine or decitabine “unmask” the gene coding for vitamin D receptors (by reducing the methylation of its promoter region). As a consequence, more vitamin D receptors are expressed and available to be activated by inecalcitol, resulting in an improved efficacy to limit leukemia progression over the hypo-methylating agents alone.
Azacytidine (Vidaza(R), Celgene) and decitabine (Dacogen (R), Janssen-Cilag) are two hypo-methylating agents already used for AML in older (>65 years old) or frail patients not eligible to standard induction chemotherapy. Inecalcitol alone has already been studied in a Phase II clinical trial in chronic lymphocytic leukemia.
Most read news
Topics
Organizations
Other news from the department research and development

Get the life science industry in your inbox
By submitting this form you agree that LUMITOS AG will send you the newsletter(s) selected above by email. Your data will not be passed on to third parties. Your data will be stored and processed in accordance with our data protection regulations. LUMITOS may contact you by email for the purpose of advertising or market and opinion surveys. You can revoke your consent at any time without giving reasons to LUMITOS AG, Ernst-Augustin-Str. 2, 12489 Berlin, Germany or by e-mail at revoke@lumitos.com with effect for the future. In addition, each email contains a link to unsubscribe from the corresponding newsletter.
More news from our other portals
Last viewed contents
QIAGEN and Roche settle dispute over distribution agreement for TheraScreen companion diagnostics

Salt could be a key factor in allergic immune reactions - Atopic dermatitis: elevated salt concentrations in affected skin

Contact lenses with medicine and sugar
Deutsches Primatenzentrum GmbH (DPZ) - Göttingen, Germany
Creabilis Raises €15M in Series B Fundraising Round
A giant called MIMAS in the cell power plants - Mega protein complex with surprising properties discovered in mitochondria
Rapid detection of antibiotic-resistant bacteria
BIOTECON Diagnostics signs license agreement with Applera Corporation
News from the Mailroom of the Cell - Researchers elucidate mechanism of protein

CureVac Starts Phase 1 Clinical Study of Modified, Omicron-Targeting COVID-19 Vaccine Candidate - Milestone demonstrates CureVac’s continued execution on comprehensive clinical program of second-generation vaccine candidates for infectious diseases
Dr. August Wolff Arzneimittel GmbH & Co. KG and Mercachem B.V. start two drug development collaborations