European Commission Grants Marketing Authorizations for AbbVie's VIEKIRAX + EXVIERA
AbbVie announced that the European Commission has granted marketing authorizations for its all-oral, short-course, interferon-free treatment of VIEKIRAX® (ombitasvir/paritaprevir/ritonavir tablets) + EXVIERA® (dasabuvir tablets). The treatment has been approved with or without ribavirin (RBV) for patients with genotype 1 (GT1) chronic hepatitis C virus (HCV) infection, including those with compensated liver cirrhosis, HIV-1 co-infection, patients on opioid substitution therapy and liver transplant recipients. Additionally, VIEKIRAX has been approved for use with RBV in genotype 4 (GT4) chronic hepatitis C patients.
"The approval of AbbVie's hepatitis C treatment in the European Union, following the recent approvals in the U.S. and Canada, offers patients across Europe a new and effective treatment to cure this serious disease," said Richard Gonzalez, chairman of the board and chief executive officer, AbbVie. "We are committed to working with local governments and healthcare systems to support broad access to VIEKIRAX + EXVIERA."
The approvals follow a review under accelerated assessment by the European Medicines Agency, designated to new medicines of major public health interest. Approximately nine million people in Europe are infected with chronic hepatitis C, a major cause of liver cancer and liver transplantation Genotype 1 is the most prevalent form of hepatitis C in Europe, accounting for 60 percent of cases worldwide. In Europe, the most prevalent sub-genotype is 1b (47 percent). Genotype 4, most common in the Middle East, sub-Saharan Africa and Egypt, is becoming increasingly prevalent in several European countries, including Italy, France, Greece and Spain.7 AbbVie's treatment is now licensed for use in all 28 member countries of the European Union, as well as in the U.S., Canada, Switzerland, Iceland, Liechtenstein and Norway.
Most read news
Organizations
Other news from the department research and development

Get the life science industry in your inbox
By submitting this form you agree that LUMITOS AG will send you the newsletter(s) selected above by email. Your data will not be passed on to third parties. Your data will be stored and processed in accordance with our data protection regulations. LUMITOS may contact you by email for the purpose of advertising or market and opinion surveys. You can revoke your consent at any time without giving reasons to LUMITOS AG, Ernst-Augustin-Str. 2, 12489 Berlin, Germany or by e-mail at revoke@lumitos.com with effect for the future. In addition, each email contains a link to unsubscribe from the corresponding newsletter.
Most read news
More news from our other portals
Last viewed contents
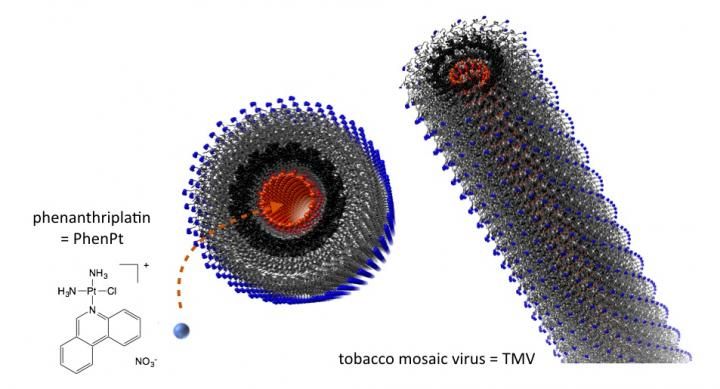
Drug candidate shrinks tumor when delivered by plant virus nanoparticle - Phenanthriplatin outperformed cisplatin in mouse model when encapsulated into nanocarrier

Tracking Down Organic Fraudsters

Zifo acquires beyontics - Comprehensive range of laboratory services & solutions





















































