Domainex Secures £1.4m Biomedical Catalyst Award
Domainex Ltd announced that its lead compound has demonstrated a more potent effect in a key model of chronic obstructive pulmonary disease (COPD) than either roflumulast or a p38 inhibitor. It has also successfully won a £1.4m Biomedical Catalyst Award to support further development towards Phase I clinical studies.
Roflumilast is a PDE4 inhibitor, known to have clinical benefit in COPD patients, and the p38 inhibitor is currently in late-stage clinical development. In the present study, Domainex compared the effect of oral doses of its compound with the same oral doses of either roflumilast or a p38 inhibitor in a COPD model that is widely-recognised to be the ‘gold standard’ pre-clinical study. The Domainex compound showed more than twice the effect of the comparator drugs in reducing the cigarette smoke-induced influx of inflammatory cells, particularly neutrophils, into the lung.
Domainex has previously shown that its inhibitors of TBK1 and IKKε are potent anti-inflammatory agents, which can inhibit signaling pathways activated by Toll-like Receptors and IL-17. This data suggests that these compounds may have utility in COPD and other inflammatory diseases including rheumatoid arthritis, lupus and psoriasis.
Dr Trevor Perrior, Research Director at Domainex, said: “These are very exciting results which suggest that our programme could lead to an oral drug for the treatment of COPD with a much better anti-inflammatory effect than existing medicines. This could provide the first truly-effective disease-modifying treatment for COPD, which would have an enormous impact on the management of this debilitating condition.”
Most read news
Other news from the department business & finance

Get the life science industry in your inbox
By submitting this form you agree that LUMITOS AG will send you the newsletter(s) selected above by email. Your data will not be passed on to third parties. Your data will be stored and processed in accordance with our data protection regulations. LUMITOS may contact you by email for the purpose of advertising or market and opinion surveys. You can revoke your consent at any time without giving reasons to LUMITOS AG, Ernst-Augustin-Str. 2, 12489 Berlin, Germany or by e-mail at revoke@lumitos.com with effect for the future. In addition, each email contains a link to unsubscribe from the corresponding newsletter.
More news from our other portals
Last viewed contents

Air pollution associated with more severe COVID-19

Artificial DNA kills cancer - Hairpin-shaped DNA binds with microRNA in cancer cells to trigger an immune response
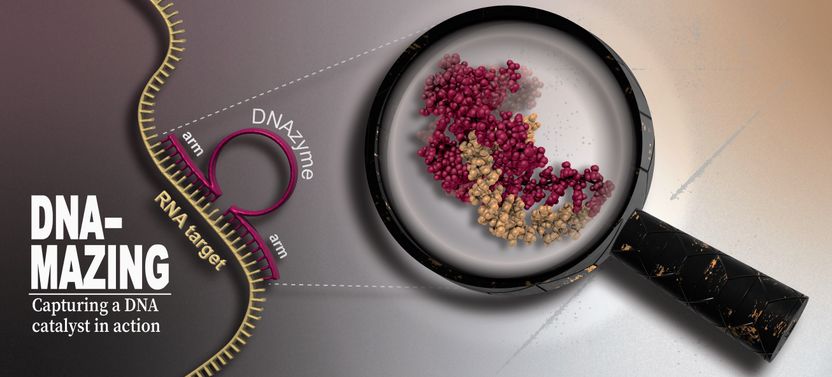
DNAzymes – how active DNA molecules with therapeutic potential work
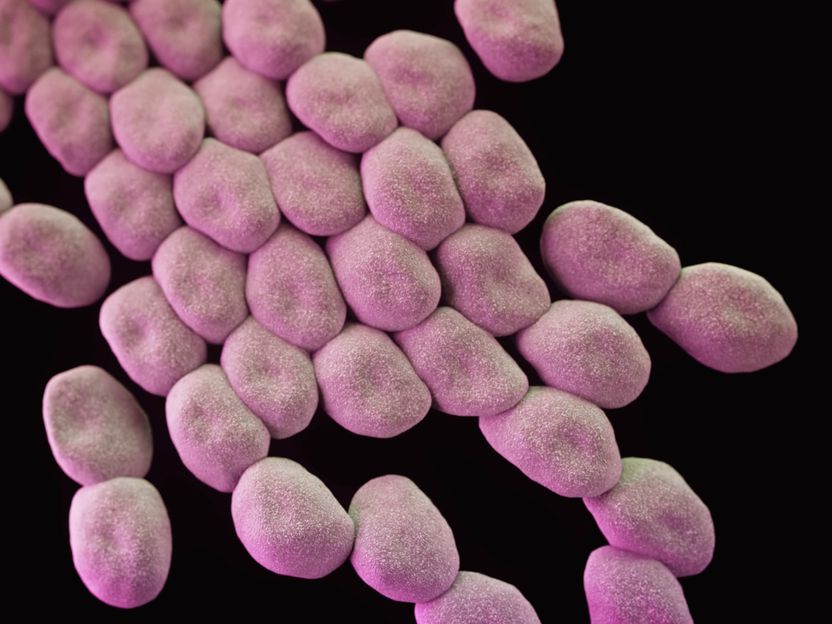
How a harmless environmental bacterium became the dreaded hospital germ Acinetobacter baumannii - And how it might possibly be combated
Scientists Find New Way to Up Safety Factor of Stem Cell Therapy by Causing Contaminated Cells to Purge Themselves
Mayo Clinic researchers discover drug can prevent colon cancer development in mice
Amgen Announces Initiation of Phase 3 Trial to Evaluate the Impact of Treating Anemia with Darbepoetin Alfa
VTT printed haemoglobin test on paper: Bioactivity presents new opportunities for paper
GENE-TRAK Listeria Microwell Test Receives AOAC Validation
MorphoSys Announces Clinical Milestone in Therapeutic Antibody Program - Seventh HuCAL Antibody to Enter Clinical Trials
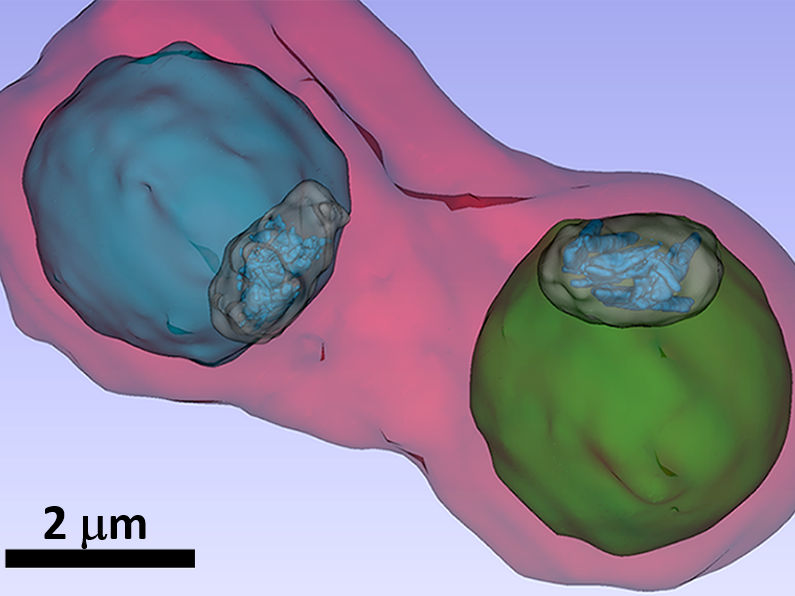
Malaria pathogens under the X-ray microscope - X-ray microscopy at BESSY II reveals how antimalaria-drugs might work























































