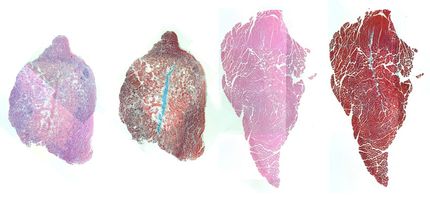
New muscular dystrophy treatment shows promise in early study
A preclinical study led by researchers in the United States has found that a new oral drug shows early promise for the treatment of muscular dystrophy. The results, which are published in EMBO Molecular Medicine, show that VBP15 decreases inflammation in mice with symptoms similar to those found in patients with Duchenne muscular dystrophy. The authors found that the drug protects and strengthens muscle without the harsh side effects linked to current treatments with glucocorticoids such as prednisone.
The researchers also observed that VBP15 inhibits the transcription factor NF-κB, a key cell-signaling molecule found in most animal cell types that plays a role in inflammation and tissue damage.
The study authors previously found out that NF-κB is active in dystrophin-deficient muscle years before the onset of symptoms, suggesting that very early treatment of Duchenne Muscular Dystrophy patients with VBP15 may prevent or delay the onset of some clinical symptoms.
“It is becoming increasingly clear that membrane integrity and repair are crucial factors in muscle, cardiovascular, neurodegenerative and airway disorders. The chemical properties of VBP15 also suggest potential for the treatment of other diseases.” remarked Kanneboyina Nagaraju, DVM, PhD, the lead author of the study and a principal investigator in the Center for Genetic Medicine Research, Children’s National Medical Center in Washington, DC.
The authors conclude that VBP15 merits further investigation for efficacy in clinical trials.
Original publication
Most read news
Original publication
Christopher R. Heier, Jesse M. Damsker, Qing Yu, Blythe C. Dillingham, Tony Huynh, Jack H. Van der Meulen, Arpana Sali, Brittany K. Miller, Aditi Phadke, et al.; "VBP15, a novel anti-inflammatory and membrane-stabilizer, improves muscular dystrophy without side effects."; EMBO Molecular Medicine
Organizations
Other news from the department science

Get the life science industry in your inbox
By submitting this form you agree that LUMITOS AG will send you the newsletter(s) selected above by email. Your data will not be passed on to third parties. Your data will be stored and processed in accordance with our data protection regulations. LUMITOS may contact you by email for the purpose of advertising or market and opinion surveys. You can revoke your consent at any time without giving reasons to LUMITOS AG, Ernst-Augustin-Str. 2, 12489 Berlin, Germany or by e-mail at revoke@lumitos.com with effect for the future. In addition, each email contains a link to unsubscribe from the corresponding newsletter.
More news from our other portals
Last viewed contents
EBE and EuropaBio hail EU Orphan Medicinal Products Regulation a success
UCI researchers find new way to fight cocaine addiction
Antiseptic resistance in bacteria could lead to next-gen plastics - Ancient protein pumps could be key to new green polymers

Biograil Secures Seed 2 Funding for Oral Delivery Device for Biologics
Artificial intelligence, hardware innovations boost confocal microscope’s performance - Enhancing the workhorse
Anemia: Baffling lab mystery leads to major red blood cell discovery
BioPhausia focuses and sells OTC products to Meda for SEK 190 million
The Biotechnology Industry over 30 Billion in Revenue - ASEBIO presented its annual report "Informe ASEBIO 2009"

Vitamin B6 may help keep COVID-19's cytokine storms at bay - Vitamin B6 may help calm cytokine storms and unclog blood clots linked to COVID-19's lethality
Born to break - Mutation causes fragile bones

"Dormant" magnetosome genes in non-magnetic bacteria discovered