Ablynx reports positive Phase I data for ALX-0061 in rheumatoid arthritis
Ablynx announced positive Phase I data from the single ascending dose part of the Phase I/II study with ALX-0061, the anti-IL-6R Nanobody, in patients with rheumatoid arthritis (RA). Based on these positive interim data, Ablynx has initiated a multiple ascending dose Phase II study with ALX-0061 in patients with RA.
During the period April to September 2011, the Phase I study recruited a total of 28 RA patients who received either placebo or a single dose of ALX-0061, injected intravenously. The single doses ranged from 0.3 mg/kg to 6 mg/kg. The study treatment was safe and well tolerated, and the biological effective doses were achieved at the doses established in the pre-clinical studies. The dose-dependent changes of the assessed biomarkers of early inflammation (C-reactive protein, fibrinogen and platelets) were consistent with the inhibition of the IL-6 pathway.
Following this study, three doses were selected for the Phase II study, including 1 mg/kg every 4 weeks, 3 mg/kg every 4 weeks, and 6 mg/kg every 8 weeks. This proof-of-concept Phase II study, which has been initiated today, is expected to recruit 36 RA patients. A first read out, including efficacy measures (DAS, ACR scores) is anticipated after 12 weeks and a final read out, including biomarker responses, is planned after 24 weeks.
Organizations
Other news from the department research and development

Get the life science industry in your inbox
By submitting this form you agree that LUMITOS AG will send you the newsletter(s) selected above by email. Your data will not be passed on to third parties. Your data will be stored and processed in accordance with our data protection regulations. LUMITOS may contact you by email for the purpose of advertising or market and opinion surveys. You can revoke your consent at any time without giving reasons to LUMITOS AG, Ernst-Augustin-Str. 2, 12489 Berlin, Germany or by e-mail at revoke@lumitos.com with effect for the future. In addition, each email contains a link to unsubscribe from the corresponding newsletter.
Most read news
More news from our other portals
Last viewed contents
Seymour_Itzkoff
The first ant methylomes uncover the relationship between DNA methylation and caste differentiation
German_Brain_Tumor_Association
Category:Flaviviruses
Endogenous_retrovirus
Phoma_exigua_var._foveata
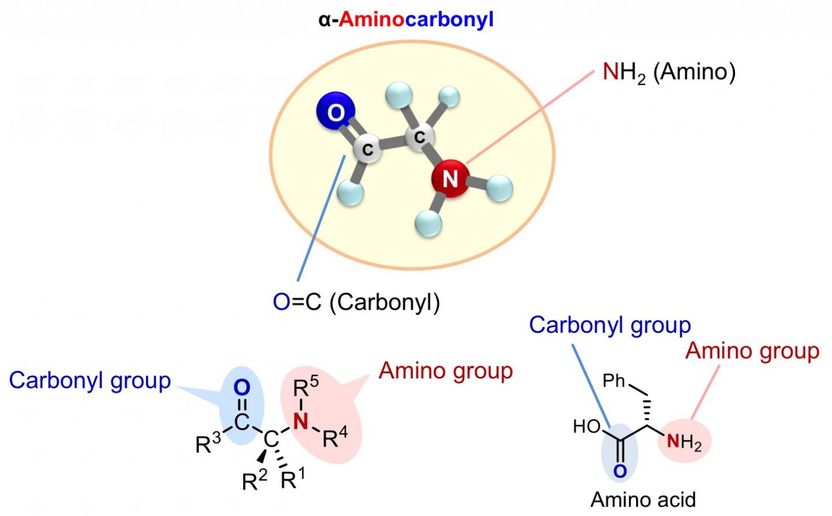
Rapid synthesis towards optically active alpha-aminocarbonyl therapeutics - New catalytic asymmetric reaction directly installs amines into carbonyl compounds
Metolazone
Category:Uncategorised_pharmacology_articles
Bothrops_lanceolatus
N-Phenethyl-4-piperidinone