p16 Immuno-cytochemistry shows superior clinical performance to HPV testing
mtm laboratories announced the publication of a large study in the American Journal of Clinical pathology that evaluated the clinical performance of the CE-labeled CINtec® Cytology kit in managing equivocal or mildly abnormal cytologic results.
The publication authored by Denton and colleagues and titled ”The Sensitivity and Specificity of p16INK4a Cytology vs. HPV Testing for Detecting High-Grade Cervical Disease in the Triage of ASC-US and LSIL Pap Cytology Results” shows that the interpretation of p16 cytology slides has the potential to provide a high sensitivity, with significantly higher specificity for the identification of high-grade CIN than High-risk Human Papillomavirus (HR-HPV) testing for the management of women with Pap cytology results categorized as ASC-US (Atypical Squamous Cells of Undetermined Significance) or LSIL (Low-grade Squamous Intraepithelial Lesion). These two Pap cytology categories are particularly difficult to interpret but cannot be dismissed as the majority of cases are benign, however, up to 15 % of women with such a cytology result harbor high-grade CIN disease, the immediate pre-cursor to cervical cancer.
The performance of p16INK4a immuno-cytochemistry (p16 Cytology) and HPV testing was analyzed on a series of 810 retrospectively collected ASC-US and LSIL cases. The diagnostic performance of each test was compared using the disease end point of biopsy confirmed high-grade disease (CIN2+). The sensitivity (i.e. ability to find established disease) of the two tests identifying CIN2+ was similar (p16 Cytology, up to 92.6% in ASC-US, and up to 92.2% in LSIL, vs. HPV testing found at 90.1% in ASC-US, and 95.7% in LSIL). However, the specificity of p16 Cytology was up to two times higher than that of HPV testing. This means that significantly less women would need to be sent on for further diagnostic follow-up, while still being able to identify those with established high-grade disease.
Bob Silverman, CEO of mtm laboratories, commented: “It is important to identify women within the screening program who are most likely to harbor high-grade disease with the least amount of intervention. The results of this study published in this peer review journal show that CINtec Cytology may set a new performance standard for the triage of both ASC-US and LSIL Cytology results.”
Original publication: Denton KJ, Bergeron C, Klement P, Trunk MJ, Keller T, Ridder R; "The Sensitivity and Specificity of p16INK4a Cytology vs HPV Testing for Detecting High-Grade Cervical Disease in the Triage of ASC-US and LSIL Pap Cytology Results."; Am J Clin Pathol 2010;134:12-21.
Other news from the department research and development

Get the life science industry in your inbox
By submitting this form you agree that LUMITOS AG will send you the newsletter(s) selected above by email. Your data will not be passed on to third parties. Your data will be stored and processed in accordance with our data protection regulations. LUMITOS may contact you by email for the purpose of advertising or market and opinion surveys. You can revoke your consent at any time without giving reasons to LUMITOS AG, Ernst-Augustin-Str. 2, 12489 Berlin, Germany or by e-mail at revoke@lumitos.com with effect for the future. In addition, each email contains a link to unsubscribe from the corresponding newsletter.
Most read news
More news from our other portals
Last viewed contents
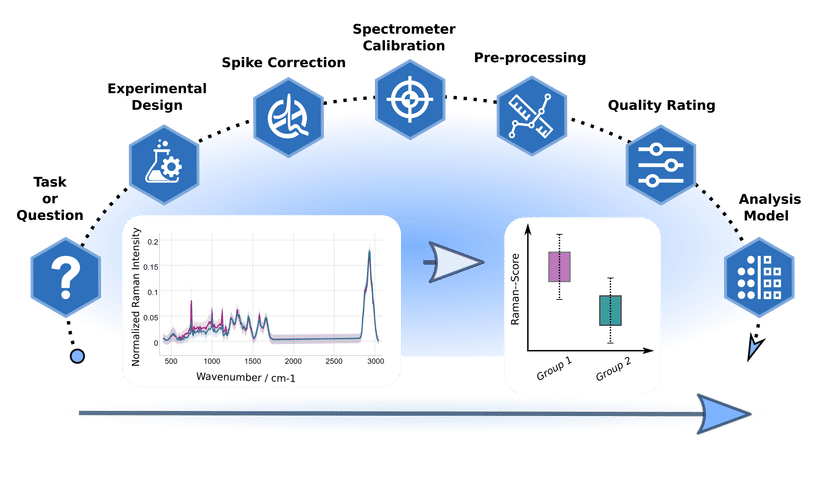
Artificial intelligence for better diagnostics - Standardized methods facilitate the evaluation of Raman spectra
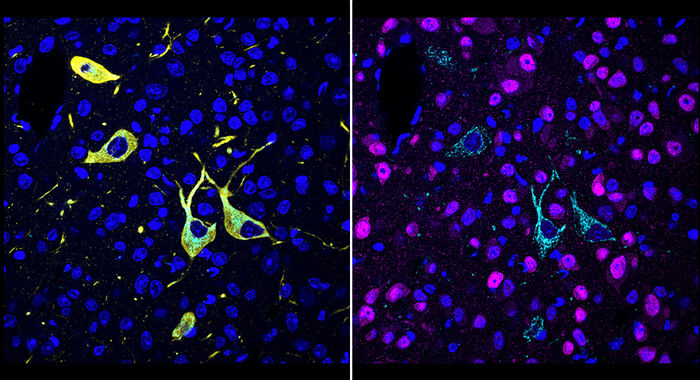
New technique shows in detail where drug molecules hit their targets in the body - The new method could speed the development of many new drugs
ChondroGene published paper ´Human genome project and cardiovascular disease genes´ - In landmark textbook ´Molecular Basis of Cardiovascular Disease´
CV Therapeutics Appoints Joseph M. Davie, M.D., Ph.D. to Board of Directors
ProtAffin AG appoints Dr. Simon Moroney to Supervisory Board
PerkinElmer Life Sciences Introduces Protein S-Nitrosylation Detection Technology - New NitroGlo(TM) Detection Kit Offers New Capabilities for Neurobiology Researchers
EPEMED the European Personalised Medicine association appoints its General Delegate and several new board members