CellCept reaches positive results in Phase III trial in Lupus Nephritis
Vifor Pharma, the pharma business sector of the Galenica Group, and Roche announced that the maintenance phase of the Aspreva Lupus Management Study (ALMS) has successfully achieved its primary endpoint. The trial demonstrates the superiority of Roche’s CellCept (mycophenolate mofetil) versus azathioprine (AZA) in delaying treatment failure in patients with Lupus nephritis (LN).
ALMS Maintenance was designed to establish the efficacy of CellCept compared to AZA as maintenance therapy in LN patients who had successfully responded to induction treatment (ALMS Induction, reported June 2007) with either CellCept or intravenous cyclophosphamide (IVC). Topline primary endpoint results for the trial indicate that CellCept was superior to AZA in delaying the time to treatment failure (p=0.003). The safety profiles of both drugs were consistent with those previously reported with no new safety signals observed.
Dr Ellen Ginzler, Chief of Rheumatology and Professor of Medicine, SUNY Downstate Medical Center New York, USA and lead investigator for the study in the US, said: “Despite recent advances in the treatment of Lupus, the disease may still relapse after successful treatment and is very difficult to study. These ALMS Study results are promising for patients who need treatment options as it has been more than 50 years since a new treatment for Lupus has been approved by the FDA.”
In light of these positive topline results, and assuming the final results are consistent, Vifor Pharma and Roche are assessing the potential to seek regulatory approval in the U.S., Europe and other major markets for the use of CellCept in the treatment of LN.
Most read news
Topics
Organizations
Other news from the department research and development

Get the life science industry in your inbox
By submitting this form you agree that LUMITOS AG will send you the newsletter(s) selected above by email. Your data will not be passed on to third parties. Your data will be stored and processed in accordance with our data protection regulations. LUMITOS may contact you by email for the purpose of advertising or market and opinion surveys. You can revoke your consent at any time without giving reasons to LUMITOS AG, Ernst-Augustin-Str. 2, 12489 Berlin, Germany or by e-mail at revoke@lumitos.com with effect for the future. In addition, each email contains a link to unsubscribe from the corresponding newsletter.
More news from our other portals
Last viewed contents
William_Potts_Dewees
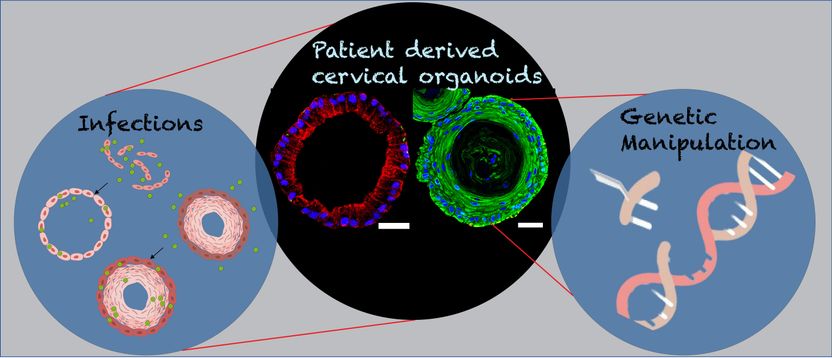
Great progress thanks to mini organs - "New opportunities are now opening up to study the biology of the cervix, infections and the development of cancer"