Single-celled kamikazes spearhead bacterial infection
How a few soldier cells confer virulence to an entire bacterial population by sacrificing themselves
Advertisement
You suddenly feel sick - pathogenic bacteria have managed to colonize and spread in your body! The weapons they use for their invasion are harmful toxins that target the host’s defense mechanisms and vital cell functions. Before these deadly toxins can attack host cells, bacteria must first export them from their production site – the cytoplasm – using dedicated secretion systems. The group of Stefan Raunser, Director at the Max Planck Institute of Molecular Physiology, has now elucidated a so-far enigmatic, exceptional secretion mechanism, that allows the release of the gigantic Tc toxins. In a kind of kamikaze attack, a small group of so-called “soldier” bacteria, packed to the brim with toxins, release their deadly cargo by exploding in the host. Targeting such subpopulations in medical therapies could be a promising treatment strategy for diseases triggered by bacteria that are becoming increasingly resistant to antibiotics.
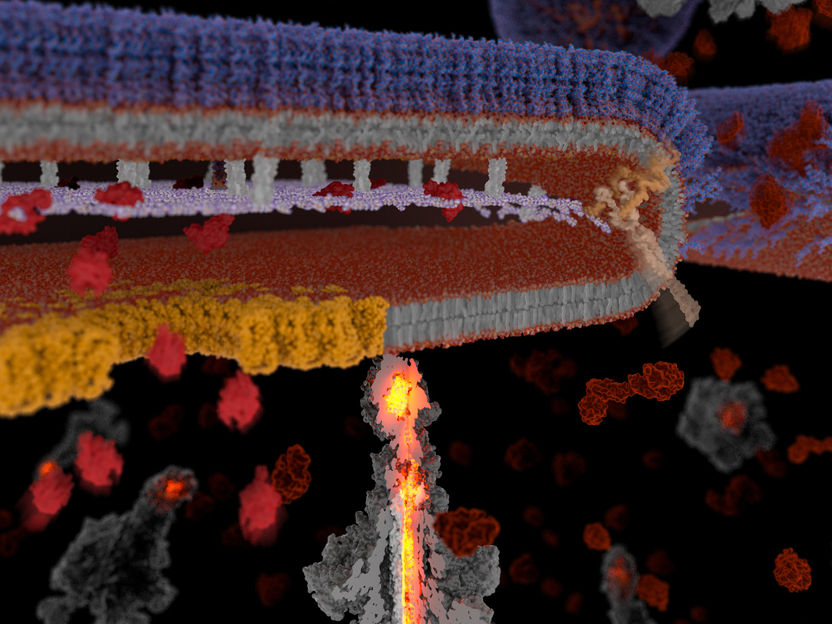
The exceptional secretion mechanism of Tc Toxins.
MPI MOPH
Once a pathogenic bacterium has entered its host, it turns on a series of defense and attack mechanisms to spread, invade and colonize deeper tissues and organs. This includes the secretion of an array of toxic proteins that subvert the host’s cellular defenses. In gram-negative bacteria, which can trigger severe infections and are becoming increasingly resistant to antibiotics, toxic proteins face the challenge of crossing several cellular barriers – belonging to both the bacteria and the host – to finally reach their destination. To this end, bacteria have developed a number of specialized secretion systems. Some can secrete a variety of toxins and are found in almost all bacteria, while others have been identified in only few bacteria. The machinery for the secretion of many smaller toxins has already been established. Not so for larger ones, like the Tc toxins produced by the notorious Yersinia bacteria, which also include pathogens that cause plague and tuberculosis. “It has remained enigmatic for decades how the huge Tc toxins reach their final destination. By obtaining the first 3D structures of a Tc toxin in our previous electron cryomicroscopy studies we could already figure out how it bypasses the last barrier, the host membrane, using a syringe-like injection mechanism. Now, we were able to complete the picture and show how these toxins overcome the three barriers separating the inside of the bacteria from its environment in a truly spectacular way,” says Stefan Raunser.
Exploding bacteria
In their recent work, Raunser and his team have applied a cutting-edge combination of several techniques to investigate the secretion of the Tc toxin YenTc produced by the insect pathogen Yersinia entomophaga, which is crucial for this bacterial species to establish an infection. The biggest challenge was to initially identify which of the known secretion machineries is used for this purpose by the bacteria. To this end, the scientists knocked out all suspected secretion systems one after the other using targeted genome editing. When none of the knockouts stopped the toxin’s release, the same technique was used to modify the toxin so that its secretion could be visualized - and this time with success. “Watching some of the bacteria literally explode to release their toxins was a real eureka moment,” says Oleg Sitsel, first author of the study. Careful proteomic analysis then finally brought to light a pH-sensitive type 10 secretion system responsible for toxin release, a class of protein export machinery that was just recently established. Subsequent cryo-electron tomographic analysis visualized the step-by-step details of how this secretion system exports cellular contents via a previously unknown lytic mode of action that overcomes the three barriers surrounding gram-negative bacteria.
Becoming a soldier cell
The scientists found that only a small specialized subset of bacterial cells produces and exports the toxins by paying the ultimate price, namely death. But what causes those cells, which the authors termed “soldier cells”, to first enlarge and produce a deadly toxin cocktail containing YenTc, then commit suicide for the benefit of their comrades? The scientists first determined that the appearance of soldier cells is temperature-, nutrient- and cell density-dependent. They then discovered a temperature-sensitive genetic switch that synchronizes the production of the toxins with the production of the secretion system, and turns “normal” cells into their soldier brethren. The mass production of toxins coupled to the cells’ enlarged size ensures that only few individuals need to be sacrificed for the greater good of the bacterial population, an extremely efficient strategy.
“We suspect that normal cells turn into soldier cells upon ingestion in response to insect host nutrients. Toxin secretion is pH sensitive, which delays its release until the soldier cells reach the alkaline posterior midgut, their major theatre of operations,” says Raunser.
“This secretion strategy is unique and remarkable. The behaviour of these bacteria exhibits characteristics such as differentiation and altruism, which are reminiscent of eusocial systems. If this turns out to be a more common mechanism, we might have exposed a weak point in bacteria: specifically targeting the soldier cells could become a promising medical strategy in the fight against pathogenic bacteria, especially in times of increasing resistance to antibiotics,” concludes Raunser.