Everything in balance? How a molecular switch controls lipid metabolism
Overactive or absent protein stops energy flow
Our body’s fat metabolism plays a vital role in energy production in our body. A research team at the University of Basel, has discovered a molecular switch that regulates lipid metabolism in our cells. This switch controls the storage or conversion of lipids into energy.
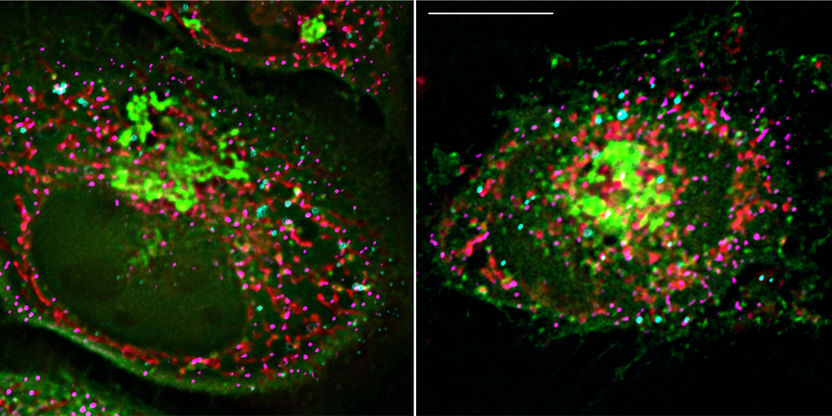
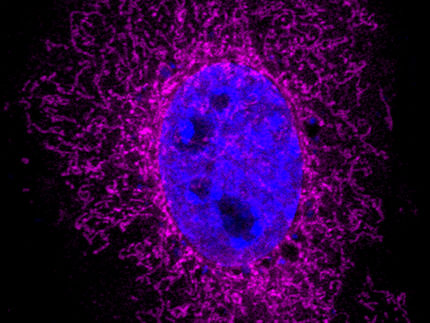
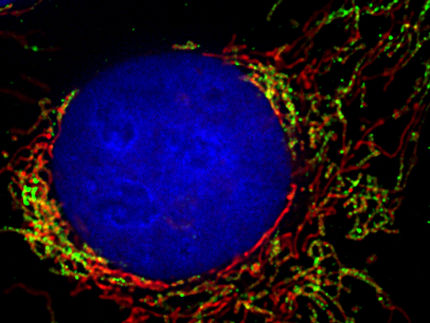
Two fluorescent microscopic images: At the contact site between mitochondrion (red) and lipid droplet (turquoise), the protein Arf1 (green) ensures that lipids are shuttled into the mitochondria, where they are converted into the energy donor ATP.
University of Basel, Biozentrum
All organisms need energy to live. We get energy from various components of our food. Our body uses a part of this energy directly and stores the rest. While glucose serves as an immediately available energy source, fats are stored as energy reserve in form of lipid droplets within our cells.
When the body needs energy from these fat stores, lipids are transported to mitochondria - the powerhouse of the cell. Here, the lipids are converted into ATP (adenosine triphosphate), a key molecule providing energy to the cells.
But how much energy does our body need from these energy stores? What proportion of lipids should be converted into ATP? When should this process start and when should it end? The research team led by Professor Anne Spang at the Biozentrum of the University of Basel has investigated lipid metabolism more closely in both yeast and human cells. The scientists discovered that a protein called Arf1 works like a molecular switch, regulating these processes. The results have recently been published in “Nature Cell Biology”.
Arf1 changes the contact site
“Arf1 is a familiar protein to us. We already know that it has several functions in the Golgi apparatus, the cell’s sorting station. We have now discovered that Arf1 also plays a role in regulating energy metabolism in the mitochondria”, explains Dr. Ludovic Enkler, first author of the study. “Arf1 ensures the transport of lipids from lipid droplets to mitochondria.” The researchers assume that Arf1 alters the environment of the contact site between the lipid droplets and mitochondria, enabling lipids to enter the mitochondria.
When the body signals a need for energy, Arf1 allows lipids to enter the mitochondria. Once the energy demand is met, the transport is stopped. “Thus, the system only works when the feedback loop of the energy requirements works,” says Ludovic Enkler.
Overactive or absent Arf1 stops energy flow
"However, if the Arf1 protein is absent or overactive, the entire system gets out of balance," explains Anne Spang. “In both cases, the feedback control between demand and production does not work, leading to insufficient ATP energy supply. Consequently, fatty acids accumulate in lipid droplets.”
The sensitivity and intricate complexity of the lipid metabolism become evident when considering various lipid metabolism disorders. Even the smallest errors in lipid metabolism can lead to high cholesterol levels (blood lipid levels), increasing the risk of cardiovascular diseases, obesity, or diabetes.
Using advanced techniques such as spatial proteomics, which can be used to study all proteins on different cellular structures, the research team aims to identify individual components involved in the feedback process of Arf1 protein in the cells. Their goal is to unravel lipid trafficking at the contact sites between lipid droplets and the mitochondria in detail.
Original publication
Ludovic Enkler, Viktoria Szentgyörgyi, Mirjam Pennauer, Cristina Prescianotto-Baschong, Isabelle Riezman, Aneta Wiesyk, Reut Ester Avraham, Martin Spiess, Einat Zalckvar, Roza Kucharczyk, Howard Riezman, and Anne Spang: Arf1 coordinates fatty acid metabolism and mitochondrial homeostasis. Nature Cell Biology (2023)