Breakthrough in the development of new anti-inflammatory therapies
Novel approach to treatment also works in patients with inflammatory bowel diseases
Advertisement
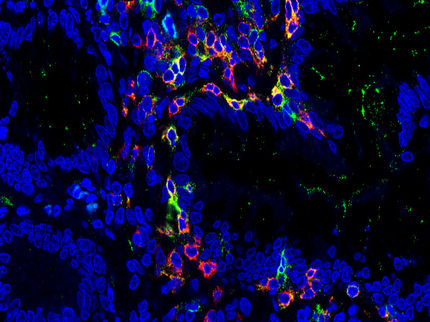
Chronic inflammatory bowel diseases (IBD), such as Crohn's disease and ulcerative colitis, lead to repeated flares of chronic diarrhea, fever and pain, as well as severe psychological stress. In Germany, around 320,000 people are affected by IBD. A disturbed immune response leads to inflammation in the gastrointestinal tract, which flares up again and again in bursts. There are only a few approved drugs for these diseases, and their efficacy is limited to a minority of patients. Therefore, there is a large unmet need for therapies to fight IBD via new mechanisms of action. Some time ago, researchers from the Cluster of Excellence "Precision Medicine in Chronic Inflammation" constructed a fusion protein that was further developed into the drug candidate "olamkicept". It suppresses bowel inflammation via a new mechanism – but this has so far only been demonstrated in different animal models. In a recent study published in the scientific journal Gastroenterology, the team led by Professor Stefan Schreiber, spokesperson of the Cluster of Excellence PMI, director of the Institute of Clinical Molecular Biology (IKMB) at Kiel University (CAU) and the University Medical Center Schleswig-Holstein (UKSH), Campus Kiel, and director of the Department of Internal Medicine I at the UKSH, Campus Kiel, have proven that olamkicept also blocks the crucial signaling pathway in patients with IBD, and can thus successfully suppress the chronic inflammation.

Prof. Dr. Stefan Schreiber, Spokesperson of theCluster of Excellence PMI, Director of the Institute for Clinical Molecular Biology, Kiel University and UKSH, and Director of the Clinic for Internal Medicine I, UKSH, Campus Kiel.
J. Haacks, Uni Kiel
New mechanism of action via the IL-6 trans-signaling pathway
Olamkicept acts via a special signaling pathway of an important chemical messenger in the immune system, which is called Interleukin-6 (IL-6). This signaling molecule is secreted in increased quantities by the body in the event of inflammation, and it is involved in the regulation of many crucial functions of the immune system. However, if a drug blocks all of the effects of IL-6, it will very successfully suppress the inflammation, but at the same time can cause severe side effects. By blocking IL-6, the immune system can be suppressed to such an extent that the body becomes significantly more susceptible to infections. This is the case with antibodies against IL-6 or its specific receptor, which are already available on the market.
“What is special about olamkicept is that it specifically inhibits only part of the IL-6 effect, via the so-called IL-6 trans-signaling pathway. Previous research suggests that the classic IL-6 signaling pathway, in particular, plays a role in immune defense, for example against pathogens. We assume that the trans-signaling pathway, in contrast, is particularly active in cases of chronic inflammation," explained co-author Professor Philip Rosenstiel, board member of the Cluster of Excellence PMI and director of the IKMB. "By specifically blocking only the trans-signaling pathway, the classic IL-6 signaling pathway, which is important for a healthy immune response, remains undisturbed," explained Rosenstiel.
In the "classic" signaling pathway, IL-6 binds to its specific receptor, which is only present on the surface of certain cells, such as immune cells or liver cells. Together, IL-6 and its specific receptor subsequently form a complex with another surface protein on the same cell, the so-called gp130 protein, which then triggers a signal in the cell. However, the specific IL-6 receptor is also present in soluble form in the blood. In the alternative IL-6 trans-signaling pathway, IL-6 binds to this soluble IL-6 receptor circulating in the blood and can then elicit a signal via the ubiquitous gp130 protein on practically all cells of the body. Thus, IL-6 trans-signaling can theoretically affect every cell in the body.
From fundamental research to drug development
Professor Stefan Rose-John, cluster member and director of the Institute of Biochemistry at the CAU, discovered this alternative signaling pathway more than 20 years ago and pioneered its research. Together with his colleagues, he developed the fusion protein "sgp130Fc" based on this research, which is the prototype of olamkicept. It captures the complex consisting of IL-6 and its soluble receptor in the blood, thereby selectively blocking the trans-signaling pathway, without disturbing the positive effects of IL-6 via the "classic" pathway.
In order to improve its efficacy and to enable large-scale production, the protein was further developed by the Kiel biotechnology company CONARIS Research Institute AG into the drug candidate olamkicept. The pharmaceutical company Ferring, together with CONARIS and the Chinese pharmaceutical company I-Mab Biopharma, are jointly developing olamkicept as a drug. The results just published are part of a larger development program, which also includes placebo-controlled efficacy trials.
Mechanism of action confirmed in patients
In numerous animal models, sgp130Fc and olamkicept have already proven their efficacy, and successfully reduced chronic inflammations without suppressing the immune system. The severe side effects of a treatment that blocks all IL-6 functions were not observed. The Kiel research team have now tested the drug on a small group of patients with ulcerative colitis or Crohn's disease for the first time, and investigated the precise effects of the drug on the human body. "Mechanistically, the substance does exactly what it is supposed to do in humans. The IL-6 trans-signaling pathway is blocked, thereby suppressing the chronic inflammation. This evidence is an important milestone in the development of the drug from laboratory to clinical use," said Schreiber.
"However, to determine how many patients will benefit from olamkicept and to what extent, as well as the possible side effects, comprehensive clinical studies are required involving a large number of patients," emphasized Schreiber. Such a double-blind and placebo-controlled phase II clinical trial to determine efficacy and tolerability in patients with ulcerative colitis has recently been conducted by Ferring and I-Mab Biopharma. The results of this trial, which are already being evaluated, are expected to be presented in July 2021 at the annual congress of the European Crohn’s and Colitis Organisation (ECCO).
Original publication
Stefan Schreiber*, Konrad Aden*, Joana P. Bernardes*, Claudio Conrad*, …Stefan Rose-John+, Georg H. Waetzig+, Philip Rosenstiel+: "Therapeutic IL-6 trans-signalling inhibition by olamkicept (sgp130Fc) in patients with active inflammatory bowel disease"; Gastroentereology; 2021; *shared first authorship. + shared last authorship