How Molecular Chaperones Dissolve Protein Aggregates Linked To Parkinson’s Disease
Researchers unravel the mechanism of amyloid fibril disaggregation
In many neurodegenerative diseases like Parkinson’s, protein aggregates form in the brain and are assumed to contribute to neuronal cell death. Yet there exists a cellular defence mechanism that counteracts these aggregates, known as amyloid fibrils, and can even dissolve fibrils already formed. This defence mechanism is based on the activity of molecular chaperones, i.e. protein folding helpers, of the heat shock protein 70 family (Hsp70). Molecular biologists from Heidelberg University and the German Cancer Research Center investigated how the Hsp70 system disaggregates amyloid fibrils of the Parkinson-specific protein α-synuclein in a test tube. The research team led by Prof. Dr Bernd Bukau expects their research results to provide new insights into how Parkinson’s disease develops and what might be done to influence it. The results were published in two articles in the journal “Nature”.
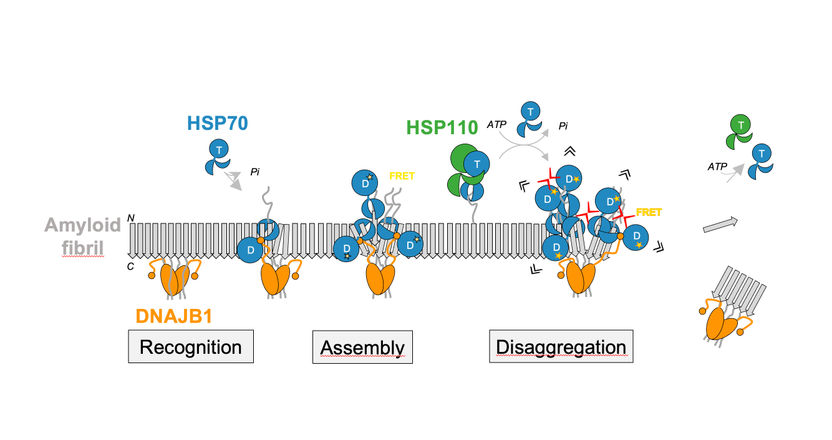
Disaggregation of α-synuclein amyloid fibrils relies on the cooperation of the HSP70 chaperone with its co-chaperones DNAJB1 and HSP110.
Bernd Bukau, Heidelberg University
Proteins in all cells – from bacteria to human – need to fold to their native state. The chains of amino acid building blocks assume specific three-dimensional structures that give proteins their functionality. This state of correct folding is constantly threatened by external and internal influences that can lead to incorrectly folded and hence damaged proteins. There is a risk that damaged proteins will “aggregate” or clump together into longer strands, the amyloid fibrils. This happens with α-synuclein in Parkinson’s disease, for example. The fibrils, in turn, are the starting point for even larger deposits.“ The formation process of such fibrillar aggregates can damage cells and even lead to cell death, as is the case in neurodegenerative diseases like Parkinson’s and Alzheimer’s,” explains Prof. Bukau, a researcher at the Center for Molecular Biology of Heidelberg University (ZMBH) and the German Cancer Research Center (DKFZ).
Prof. Bukau’s research focuses on how these protein aggregates can be dissolved. In earlier work, he and his team succeeded in identifying a cellular activity that plays a vital role in dissolving fibrillar aggregates, which is based on chaperones of the Hsp70 family. Hsp70 chaperones help other proteins with folding and can even isolate and refold aggregated proteins. The latest research by Prof. Bukau and postdoc Dr Anne Wentink shows the effects the Hsp70 chaperones have on the Parkinson-specific amyloid fibrils of the α-synuclein protein. α-synuclein is a small protein that helps in the release of messengers called neurotransmitters in the brain, although its exact function remains unclear. It became known because massive deposits of this specific protein were found in Parkinson’s patients and has been causally linked to the disease.
In biochemical experiments, the Heidelberg scientists were recently able to show that the human Hsp70 chaperone relies on the assistance of two specific co-chaperone partners to dissolve the amyloid fibrils of the α-synuclein protein. A precisely regulated interaction of these proteins leads to the formation of chaperone complexes on the surface of the fibrils, which then break up the aggregates. “It is the sheer local accumulation of many chaperone proteins on the surface of the α-synuclein fibrils that generates the force to break up the fibrils and detach the α-synuclein molecules,” explains Dr Wentink. The close proximity between the chaperones on the restricted surface of the fibrils plays a decisive role in creating strong enough pulling forces to disrupt the fibrils.
The experiments were conducted together with colleagues from the European Molecular Biology Laboratory (EMBL) in Heidelberg, the Center for Structural Biology of Montpellier (France) and the École Polytechnique Fédérale de Lausanne (Switzerland). The project was funded by the “Top Research” programme of the Baden-Württemberg Foundation, the German Research Foundation, and the Helmholtz Association.
The second study published in “Nature” focuses on a previously unknown regulatory mechanism, a type of molecular switch that sets in motion the overall Hsp70 chaperone activity to dissolve the amyloid fibrils. This mechanism is based on a sequence of direct interactions between the different parts of the DNAJB1 co-chaperone and the Hsp70 chaperone. This ultimately activates Hsp70 to use ATP as an energy source, making it possible to productively bind to the fibrils and effect their disintegration. A close research collaboration of Dr Rina Rosenzweig of the Weizman Institute of Science in Rehovot (Israel) and Prof. Bukau, Dr Wentink and Dr Nadinath Nillegoda of Monash University in Melbourne (Australia) – a former Alexander von Humboldt Foundation fellow in Bernd Bukau’s research group – succeeded in identifying this mechanism.
“Our latest results from the two studies give us a molecular understanding of how amyloid fibrils are dissolved. We were able to demonstrate that the chaperones work like a machine to dissolve the fibrils,” adds Prof. Bukau. According to the Heidelberg researcher, this opens up new avenues for the development of agents that specifically target the chaperone-based cellular defence mechanism against amyloid formation. A better understanding of how this chaperone activity influences the course of neurodegenerative diseases will thus be of essential importance in the therapeutic exploitation of the findings described in these studies.
Original publication
A.S. Wentink, N.B. Nillegoda, J. Feufel, G. Ubartait, C.P. Schneider, P. De Los Rios, J. Hennig, A. Barducci, B. Bukau; "Molecular dissection of amyloid disaggregation by the human Hsp70 chaperone"; Nature; 2020.
O. Faust, M. Abayev-Avraham, A.S. Wentink, M. Maurer, N.B. Nillegoda, N. London, B. Bukau, R. Rosenzweig; "Hsp40s employ class-specific regulation to drive Hsp70 functional diversity"; Nature; 2020