YM BioSciences announces early expansion of ongoing CYT387 Phase I/II clinical study based on favorable safety and activity data
YM BioSciences Inc. announced that it has received ethics board approval to expand enrolment in its Phase I/II clinical trial of CYT387 at Mayo Clinic in patients with myelofibrosis, a chronic debilitating condition, where patient's bone marrow is replaced by scar tissue.
"The favorable safety and biological activity data we have collected to date in this study gave us the confidence to seek approval for cohort expansion earlier than originally contemplated," said Dr. Ayalew Tefferi, Professor of Hematology at Mayo Graduate School and Chair of the study. Enrolment expansion will facilitate the collection of more safety, tolerability and preliminary efficacy data and may assist with planning for subsequent registration-enabling clinical studies for patients with myelofibrosis and other myeloproliferative neoplasms (MPNs).
"These preliminary findings also advance the prospect for more rapid initiation of subsequent clinical programs," said David Allan, Chairman & CEO of YM BioSciences. "The JAK2 inhibitors, including CYT387, are of great interest to the global pharmaceutical industry. They hold therapeutic promise in numerous indications. Myelofibrosis alone is a disease that affects approximately 20,000 patients in North America with market estimates in excess of $750 million."
Enrolment into the Company's Phase I/II study with myelofibrosis commenced in November 2009 at Mayo Clinic, Rochester, MN. Phase II efficacy data for CYT387 were originally anticipated in the second half of 2011, however the evident safety and preliminary efficacy observed to date support early expansion and should allow conclusion of the study three to six months earlier. This in turn may enable more rapid selection of doses for registration-enabling Phase III studies in myelofibrosis. Enrolment of approximately 60 patients was originally planned across both phases of the study, with the majority of patients to be enrolled in the later Phase II portion. However, cohort expansion will allow for more patients to be dosed during 2010, bringing forward the possibility of a rapid progression into an NDA-enabling study in myelofibrosis as well as other Phase II studies in other hematology and oncology indications with unmet medical needs.
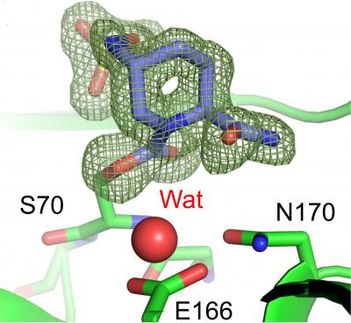
CYT387 is a potent inhibitor of the kinase enzymes JAK1 and JAK2, which have been implicated in a family of hematological conditions known as myeloproliferative neoplasms, including myelofibrosis. Typical myelofibrosis symptoms include an enlarged spleen, progressive anemia and poor overall survival.
Most read news
Other news from the department research and development

Get the life science industry in your inbox
By submitting this form you agree that LUMITOS AG will send you the newsletter(s) selected above by email. Your data will not be passed on to third parties. Your data will be stored and processed in accordance with our data protection regulations. LUMITOS may contact you by email for the purpose of advertising or market and opinion surveys. You can revoke your consent at any time without giving reasons to LUMITOS AG, Ernst-Augustin-Str. 2, 12489 Berlin, Germany or by e-mail at revoke@lumitos.com with effect for the future. In addition, each email contains a link to unsubscribe from the corresponding newsletter.