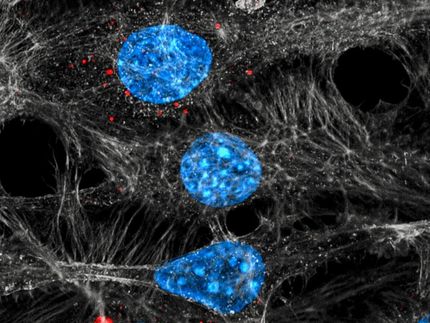
Herceptin now approved in the EU for patients with HER2-positive advanced stomach cancer
First targeted biological therapy to show survival benefit in stomach cancer
Roche announced that the European Commission has approved Herceptin (trastuzumab) in combination with chemotherapy for use in patients with HER2-positive metastatic stomach (gastric) cancer. The approval is based on the results from the international ToGA trial, which showed that treatment with Herceptin significantly prolongs the lives of patients with this aggressive cancer. Overall survival for patients with high levels of HER2 in the ToGA study was 16 months versus 11.8 months (on average) for patients receiving chemotherapy alone.
“Herceptin is the first targeted biological therapy to show a survival benefit in advanced stomach cancer and represents a significant advance in the treatment of this devastating disease”, said Pascal Soriot, Chief Operating Officer (COO), Roche Pharmaceutical Division. “We believe that Herceptin will help patients with HER2-positive stomach cancer, as much as it has helped so many women with HER2-positive breast cancer.”
Based on the strong results from the phase III ToGA study, the submission for the label extension was reviewed in an accelerated process by the European Health Authorities, allowing patients to benefit sooner from this life-extending treatment. This marketing authorisation is valid with immediate effect in all European Union (EU) and EEA-EFTA states (Iceland, Liechtenstein and Norway). Following approval in the European Union, approvals for a label extension for Herceptin in other regions of the world are expected to follow soon.
ToGA is the first randomised Phase III trial investigating the use of Herceptin in patients with inoperable locally advanced, recurrent and/or metastatic HER2-positive stomach cancer. Approximately 3,800 patients were tested for HER2-positive tumours and 594 patients with HER2-positive disease were enrolled into the study. The rationale for conducting this trial was based on the knowledge that the targeted therapy Herceptin has demonstrated unprecedented efficacy in the treatment of HER2-positive breast cancer. In addition, the overexpression of HER2 was also observed in stomach cancer. Targeted cancer therapies are drugs or other substances that block the growth and spread of cancer by interfering with specific molecules involved in tumour growth and progression.
Most read news
Topics
Organizations
Other news from the department research and development

Get the life science industry in your inbox
By submitting this form you agree that LUMITOS AG will send you the newsletter(s) selected above by email. Your data will not be passed on to third parties. Your data will be stored and processed in accordance with our data protection regulations. LUMITOS may contact you by email for the purpose of advertising or market and opinion surveys. You can revoke your consent at any time without giving reasons to LUMITOS AG, Ernst-Augustin-Str. 2, 12489 Berlin, Germany or by e-mail at revoke@lumitos.com with effect for the future. In addition, each email contains a link to unsubscribe from the corresponding newsletter.
Most read news
More news from our other portals
Last viewed contents
Silence Therapeutics Forms Scientific Advisory Board
InteRNA Technologies and Dana-Farber Cancer Institute to collaborate on the role of microRNAs in cancer pathways
Bayer Animal Health and Paraco agree on access to lead molecules
Wiggly proteins guard the genome - Dynamic network in the pores of the nuclear envelope blocks dangerous invaders

Defective immune cells in the brain cause Alzheimer’s disease - Scientists are studying the role of immune cell activation in Alzheimer's disease

Sepsis in Children: Improved Diagnosis Thanks to New Global Criteria - Researchers applied machine-learning methods to extrapolate from the data analysis evidence-based criteria for diagnosing